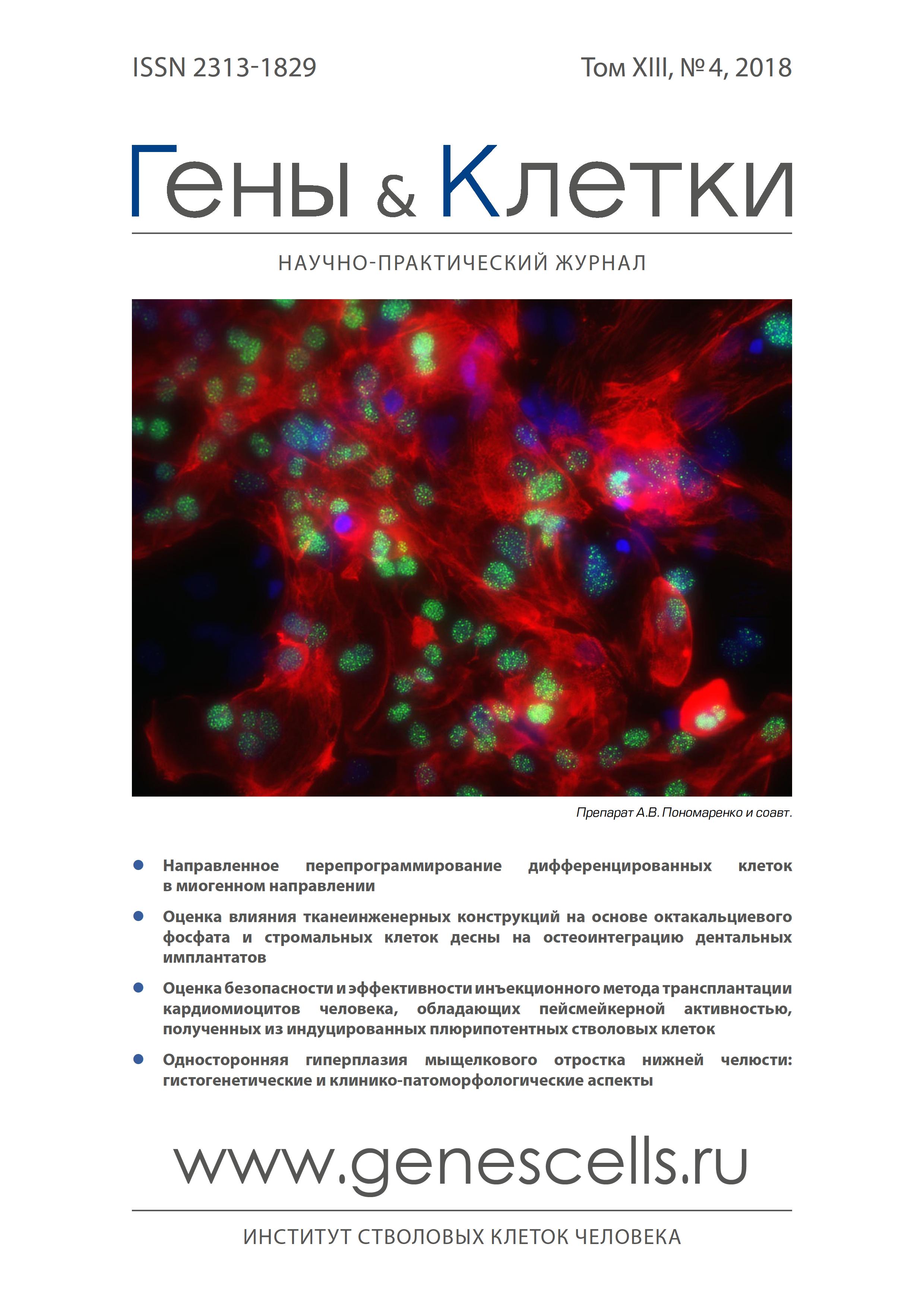
Evaluation of the effect of tissue-engineered constructs based on octacalcium phosphate and gingival stromal cells on dental implants osteointegration
- Authors: Bozo I.Y1,2,3, Deev R.V1,3,4,5, Volkov A.V6, Eremin I.I3, Korsakov I.N3, Yasinovsky M.I3, Ustinov K.D3, Trofimov V.O1,2, Ruzin I.7, Presnyakov E.V4, Komlev V.S8, Pulin A.A3
-
Affiliations:
- Histograft, LLC
- A.I. Burnazyan Federal Medical and Biophysical Center, FMBA Russia
- Research Institute of General Pathology and Pathophysiology
- I.P. Pavlov Ryazan State Medical University
- PJSC «Institute of Human Stem Cells»
- E.O. Mukhin City Clinical Hospital № 70
- A.I. Evdokimov Moscow State University of Medicine and Dentistry
- AA. Baykov Institute of Metallurgy and Materials Science of the RAS
- Issue: Vol 13, No 4 (2018)
- Pages: 24-30
- Section: Articles
- Submitted: 05.01.2023
- Published: 15.12.2018
- URL: https://genescells.ru/2313-1829/article/view/120719
- DOI: https://doi.org/10.23868/201812043
- ID: 120719
Cite item
Abstract
Full Text
About the authors
I. Y Bozo
Histograft, LLC; A.I. Burnazyan Federal Medical and Biophysical Center, FMBA Russia; Research Institute of General Pathology and Pathophysiology
Email: bozo.ilya@gmail.com
R. V Deev
Histograft, LLC; Research Institute of General Pathology and Pathophysiology; I.P. Pavlov Ryazan State Medical University; PJSC «Institute of Human Stem Cells»
A. V Volkov
E.O. Mukhin City Clinical Hospital № 70
I. I Eremin
Research Institute of General Pathology and Pathophysiology
I. N Korsakov
Research Institute of General Pathology and Pathophysiology
M. I Yasinovsky
Research Institute of General Pathology and Pathophysiology
K. D Ustinov
Research Institute of General Pathology and Pathophysiology
V. O Trofimov
Histograft, LLC; A.I. Burnazyan Federal Medical and Biophysical Center, FMBA Russia
IA. Ruzin
A.I. Evdokimov Moscow State University of Medicine and Dentistry
E. V Presnyakov
I.P. Pavlov Ryazan State Medical University
V. S Komlev
AA. Baykov Institute of Metallurgy and Materials Science of the RAS
A. A Pulin
Research Institute of General Pathology and Pathophysiology
References
- Müller A., Hussein K. Meta-analysis of teeth from European populations before and after the 18th century reveals a shift towards increased prevalence of caries and tooth loss. Arch. Oral Biol. 2017; 73: 7-15.
- Annual Report 2017. Straumann Group. https://www.straumann. com/group/en/discover/annualreport/2017.html. Дата посещения: 01.12.2018.
- Brânemark P.I., Hansson B.O., Adell R. et al. Osseointegrated implants in the treatment of the edentulous jaw. Experience from a 10-year period. Scand. J. Plast. Reconstr. Surg. Suppl. 1977; 16: 1-132.
- Attard N.J., Zarb G.A. Immediate and early implant loading protocols: a literature review of clinical studies. J. Prosthet. Dent. 2005; 94(3): 242-58.
- Morton D., Ganeles J. Протоколы протезирования в имплантологической стоматологии. Международная группа по имплантологии. Руководство по имплантологии, Том 2. ООО «Издательство «Квинтэссенция»: Москва, 2011.
- Schroeder A., Pohler O., Sutter F. Tissue reaction to an implant of a titanium hollow cylinder with a titanium surface spray layer. SSO Schweiz Monatsschr Zahnheilkd. 1976; 86(7): 713-27.
- Brânemark P.I. Osseointegration and its experimental background. J. Prosthet. Dent. 1983; 50(3): 399-410.
- Checchi V., Felice P., Zucchelli G. et al. Wide diameter immediate post-extractive implants vs delayed placement of normal-diameter implants in preserved sockets in the molar region: 1-year post-loading outcome of a randomised controlled trial. Eur. J. Oral Implantol. 2017; 10(3): 263-78.
- Weigl P., Strangio A. The impact of immediately placed and restored single-tooth implants on hard and soft tissues in the anterior maxilla. Eur. J. Oral. Implantol. 2016; 9 (Suppl 1): S89-106.
- Komlev V.S., Barinov S.M., Bozo 1.1. et al. Bioceramics composed of octacalcium phosphate demonstrate enhanced biological behavior. ACS Appl Mater Interfaces. 2014; 6(19): 16610-20.
- Бозо И.Я., Деев Р.В., Дробышев А.Ю. и др. Эффективность генактивированного остеопластического материала на основе октакальциевого фосфата и плазмидной днк с геном VEGF в восполнении «критических» костных дефектов. Вестник травматологии и ортопедии им. Н.Н. Приорова 2015; 1: 35-42.
- Деев Р.В., Дробышев А.Ю., Бозо И.Я. Ординарные и активированные остеопластические материалыВестник травматологии и ортопедии им. Н.Н. Приорова 2015; 1: 51-69.
- Деев Р.В., Дробышев А.Ю., Бозо И.Я. и др. Создание и оценка биологического действия ген-активированного остеопластического материала, несущего ген VEGF человека. Клеточная трансплантология и тканевая инженерия 2013; 8(3): 78-85.
- Деев Р.В., Цупкина Н.В., Бозо И.Я. и др. Тканеинженерный эквивалент кости: методологические основы создания и биологические свойства. Клеточная трансплантология и тканевая инженерия 2011; 6(1): 62-7.
- Зорин В.Л., Зорина А.И., Еремин И.И. и др. Сравнительный анализ остеогенного потенциала мультипотентных мезенхимальных стромальных клеток слизистой оболочки полости рта и костного мозга. Гены и Клетки; 2014; 9(1): 50-57.
- Ribeiro M., Fraguas E.H., Brito K.I.C. et al. Bone autografts & allografts placed simultaneously with dental implants in rabbits. J Craniomaxillofac Surg. 2018; 46(1): 142-7.
- ISO 10993-6:2007. Biological evaluation of medical devices - Part 6: Tests for local effects after implantation.
- Anada T., Kumagai T., Honda Y. et al. Dose-dependent osteogenic effect of octacalcium phosphate on mouse bone marrow stromal cells. Tissue Eng. Part A. 2008; 14(6): 965-78.
- Felice P., Pistilli R., Barausse C. et al. Immediate non-occlusal loading of immediate post-extractive versus delayed placement of single implants in preserved sockets of the anterior maxilla: 1-year post-loading outcome of a randomised controlled trial. Eur. J Oral Implantol. 2015; 8(4): 361-72.
Supplementary files