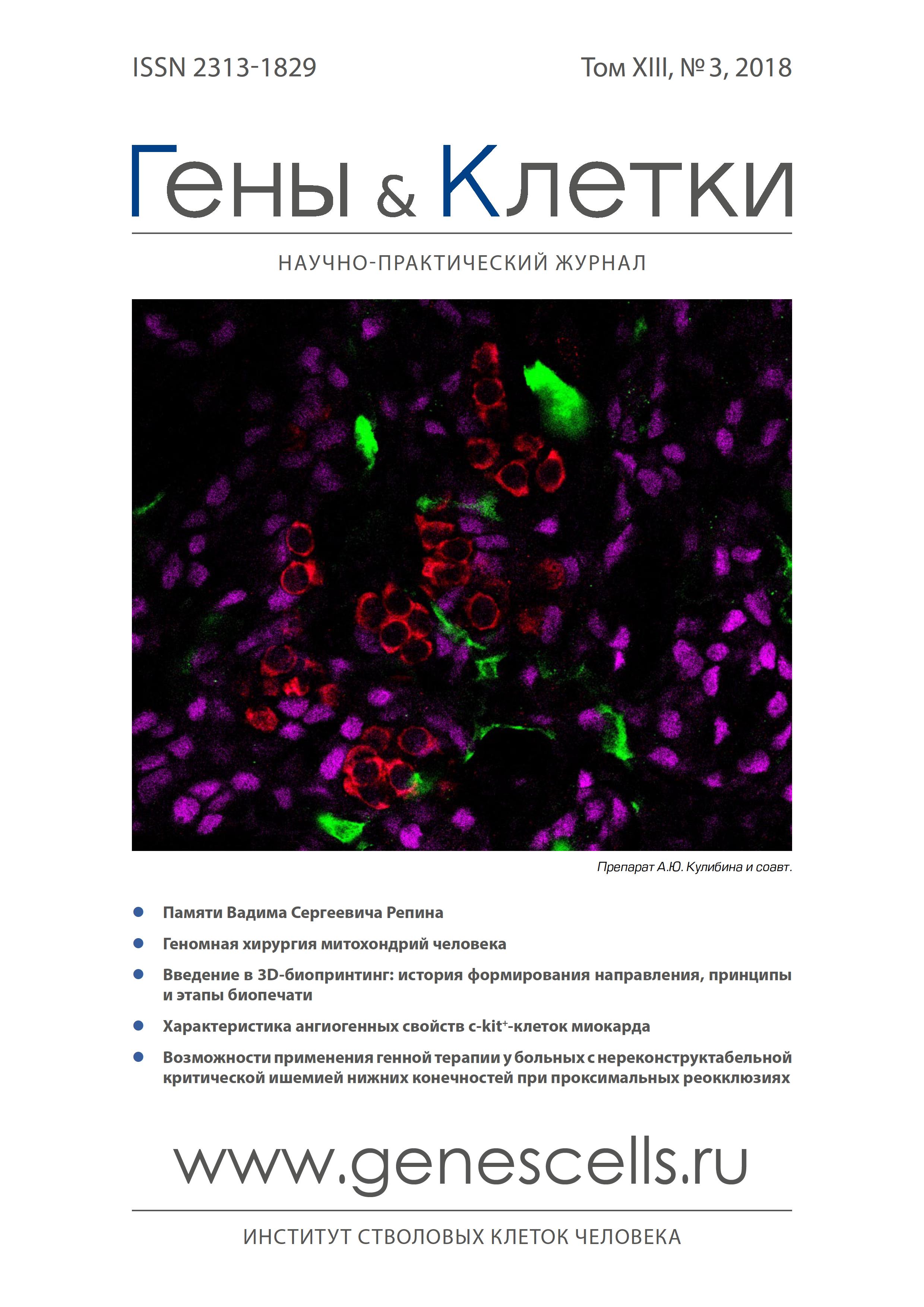
A study of physical and biological properties of 3D matrices made from polytrimethylene carbonate and its copolymers
- Authors: Chernonosova V.S1,2, Gostev A.1, Kharkova M.V1,2, Pokushalov E.1, Karpenko A.A1, Karaskov A.M1, Laktionov P.P1,2
-
Affiliations:
- E.N. Meshalkin National Medical Research Center, Ministry of Health of the Russian Federation
- Institute of Chemical Biology and Fundamental Medicine,Siberian Branch of the RAS
- Issue: Vol 13, No 3 (2018)
- Pages: 63-69
- Section: Articles
- URL: https://genescells.ru/2313-1829/article/view/120739
- DOI: https://doi.org/10.23868/201811035
- ID: 120739
Cite item
Abstract
Full Text
About the authors
V. S Chernonosova
E.N. Meshalkin National Medical Research Center, Ministry of Health of the Russian Federation; Institute of Chemical Biology and Fundamental Medicine,Siberian Branch of the RAS
AA. Gostev
E.N. Meshalkin National Medical Research Center, Ministry of Health of the Russian Federation
M. V Kharkova
E.N. Meshalkin National Medical Research Center, Ministry of Health of the Russian Federation; Institute of Chemical Biology and Fundamental Medicine,Siberian Branch of the RAS
EA. Pokushalov
E.N. Meshalkin National Medical Research Center, Ministry of Health of the Russian Federation
AA. A Karpenko
E.N. Meshalkin National Medical Research Center, Ministry of Health of the Russian Federation
A. M Karaskov
E.N. Meshalkin National Medical Research Center, Ministry of Health of the Russian Federation
P. P Laktionov
E.N. Meshalkin National Medical Research Center, Ministry of Health of the Russian Federation; Institute of Chemical Biology and Fundamental Medicine,Siberian Branch of the RAS
References
- Shaunak S., Dhinsa B., Khan W. The role of 3D modelling and printing in orthopaedic tissue engineering: A review of the current literature. Current stem cell research & therapy 2017; 12(3): 225-32.
- Husain S.R., Ohya Y., Puri R.K. Current status and challenges of three-dimensional modeling and printing of tissues and organs. Tissue Engineering Part A 2017; 23(11-12): 471-3.
- Hajiali F., Tajbakhsh S., Shojaei A. Fabrication and properties of polycaprolactone composites containing calcium phosphate-based ceramics and bioactive glasses in bone tissue engineering: a review. Polymer Reviews 2018; 58(1): 164-207.
- Asghari F., Samiei M., Adibkia K. et al. Biodegradable and biocompatible polymers for tissue engineering application: a review. Artificial cells, nanomedicine, and biotechnology 2017; 45(2): 185-92.
- Покровский А.В., Ивандаев А.С. Общее число артериальных реконструкций. В: Покровский А.В., Ивандаев А.С., редакторы. Состояние сосудистой хирургии в России в 2016 году. Москва: Российское общество ангиологов и сосудистых хирургов; 2017. с. 5-26.
- Huang Z.M., Zhang Y.Z., Kotaki M. et al. A review on polymer nano-fibers by electrospinning and their applications in nanocomposites. Composites science and technology 2003; 63(15): 2223-53.
- Pham Q.P., Sharma U., Mikos A.G. Electrospinning of polymeric nanofibers for tissue engineering applications: a review. Tissue engineering 2006; 12(5): 1197-211.
- Teo W.E., Ramakrishna S. A review on electrospinning design and nanofibre assemblies. Nanotechnology 2006; 17(14): R89.
- Li M., Mondrinos M.J., Gandhi M.R. et al. Electrospun protein fibers as matrices for tissue engineering. Biomaterials 2005; 26(30): 5999-6008.
- Taepaiboon P., Rungsardthong U., Supaphol P. Drug-loaded electrospun mats of poly (vinyl alcohol) fibres and their release characteristics of four model drugs. Nanotechnology 2006; 17(9): 2317.
- Yin A., Zhang K., McClure M.J. et al. Electrospinning collagen/chitosan/poly (L-lactic acid-co-e-caprolactone) to form a vascular graft: Mechanical and biological characterization. Journal of biomedical materials research Part A 2013; 101(5): 1292-301.
- Huang C., Chen R., Ke Q. et al. Electrospun collagen-chitosan-TPU nanofibrous scaffolds for tissue engineered tubular grafts. Colloids and Surfaces B: Biointerfaces 2011; 82(2): 307-15.
- Al-Salihi M. Electrospun Small-Diameter Silk Fibroin Vascular Grafts with Tuned Mechanical and Biocompatibility Properties as Tissue Engineered Scaffolds [Doctoral dissertation]. Baltimore [MD]: University of Maryland; 2017.
- Marcolin C., Draghi L., Tanzi M. et al. Electrospun silk fibroin-gelatin composite tubular matrices as scaffolds for small diameter blood vessel regeneration. Journal of Materials Science: Materials in Medicine 2017; 28(5): 80.
- Hasan A., Memic A., Annabi N. et al. Electrospun scaffolds for tissue engineering of vascular grafts. Acta biomaterialia 2014; 10(1): 11-25.
- Xiang P., Wang S.S., He M. et al. The in vitro and in vivo biocompatibility evaluation of electrospun recombinant spider silk protein/PCL/gelatin for small caliber vascular tissue engineering scaffolds. Colloids and Surfaces B: Biointerfaces 2018; 163: 19-28.
- Wang K., Zhang Q., Zhao L. et al. Functional Modification of Electrospun Poly (є-caprolactone) Vascular Grafts with the Fusion Protein VEGF-HGFI Enhanced Vascular Regeneration. ACS applied materials & interfaces 2017; 9(13): 11415-27.
- Madhavan K., Frid M.G., Hunter K. et al. Development of an electrospun biomimetic polyurea scaffold suitable for vascular grafting. Journal of Biomedical Materials Research Part B: Applied Biomaterials 2018; 106(1): 278-90.
- Wang X., Ding B., Li B. Biomimetic electrospun nanofibrous structures for tissue engineering. Materials Today 2013; 16(6): 229-41.
- Попова И.В., Степанова А.О., Сергеевичев Д.С. и др. Сравнительное исследование трех типов протезов, изготовленных методом электроспиннинга в эксперименте in vitro и in vivo. Патология кровообращения и кардиохирургия 2015; 19(4): 7-15.
- Pêgo A.P., Poot A.A., Grijpma D.W. et al. Physical properties of high molecular weight 1, 3-trimethylene carbonate and D, L-lactide copolymers. Journal of Materials Science: Materials in Medicine 2003; 14(9): 767-73.
- Albertsson A.C., Eklund M. Influence of molecular structure on the degradation mechanism of degradable polymers: In vitro degradation of poly (trimethylene carbonate), poly (trimethylene carbonate-co-caprolactone), and poly (adipic anhydride). Journal of applied polymer science 1995; 57(1): 87-103.
- Jaffe E.A., Nachman R.L., Becker C.G. et al. Culture of human endothelial cells derived from umbilical veins. Identification by morphologic and immunologic criteria. The Journal of clinical investigation 1973; 52(11): 2745-56.
- Feng L., Stern D.M., Pile-Spellman J. Human endothelium: endovascular biopsy and molecular analysis. Radiology 1999; 212(3): 655-64.
- Trinca R.B., Abraham G.A., Felisberti M.I. Electrospun nanofibrous scaffolds of segmented polyurethanes based on PEG, PLLA and PTMC blocks: physico-chemical properties and morphology. Materials Science and Engineering: C 2015; 56: 511-7.
- Kucinska-Lipka J., Gubanska I., Janik H. et al. Fabrication of polyurethane and polyurethane based composite fibres by the electrospinning technique for soft tissue engineering of cardiovascular system. Materials Science and Engineering: C 2015; 46: 166-76.
- Zhang Y., Ouyang H., Lim C. T. et al. Electrospinning of gelatin fibers and gelatin/PCL composite fibrous scaffolds. Journal of Biomedical Materials Research Part B: Applied Biomaterials 2005; 72(1): 156-65.
- Chernonosova V.S., Kvon R.I., Stepanova A.O. et al. Human serum albumin in electrospun PCL fibers: structure, release, and exposure on fiber surface. Polymers for Advanced Technologies 2017; 28(7): 819-27.
- Sell S.A., Wolfe P.S., Garg K. et al. The use of natural polymers in tissue engineering: a focus on electrospun extracellular matrix analogues. Polymers 2010; 2(4): 522-53.
- Liu H., Ding X., Zhou G. et al. Electrospinning of nanofibers for tissue engineering applications. Journal of Nanomaterials 2013; 2013: 3.
- Grasl C., Bergmeister H., Stoiber M. et al. Electrospun polyurethane vascular grafts: in vitro mechanical behavior and endothelial adhesion molecule expression. Journal of Biomedical Materials Research Part A 2010; 93(2): 716-23.
- Попова И.В., Степанова А.О., Плотникова Т.А. и соавт. Изучение проходимости сосудистого протеза, изготовленного методом электроспиннинга. Ангиология и сосудистая хирургия 2015; 21(2): 136-42.
- Chen Q., Liang S., Thouas G.A. Elastomeric biomaterials for tissue engineering. Progress in polymer science 2013; 38(3-4): 584-671.
Supplementary files